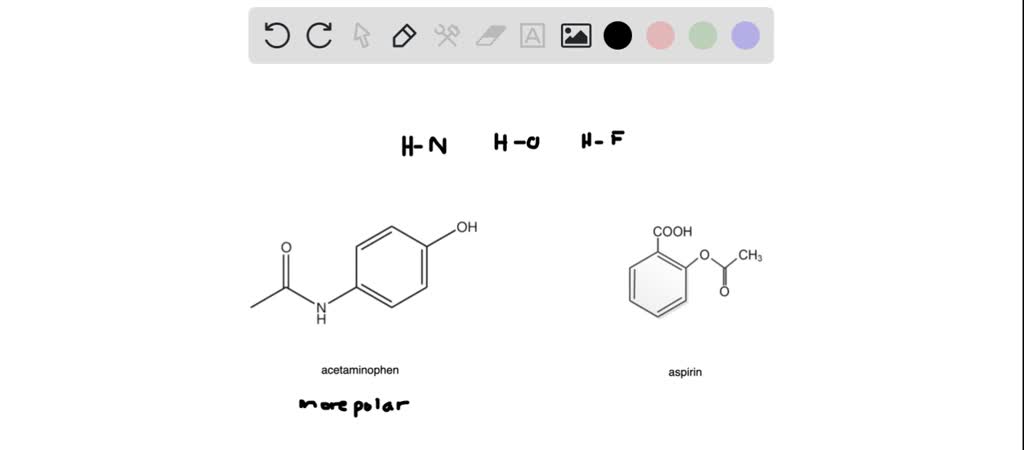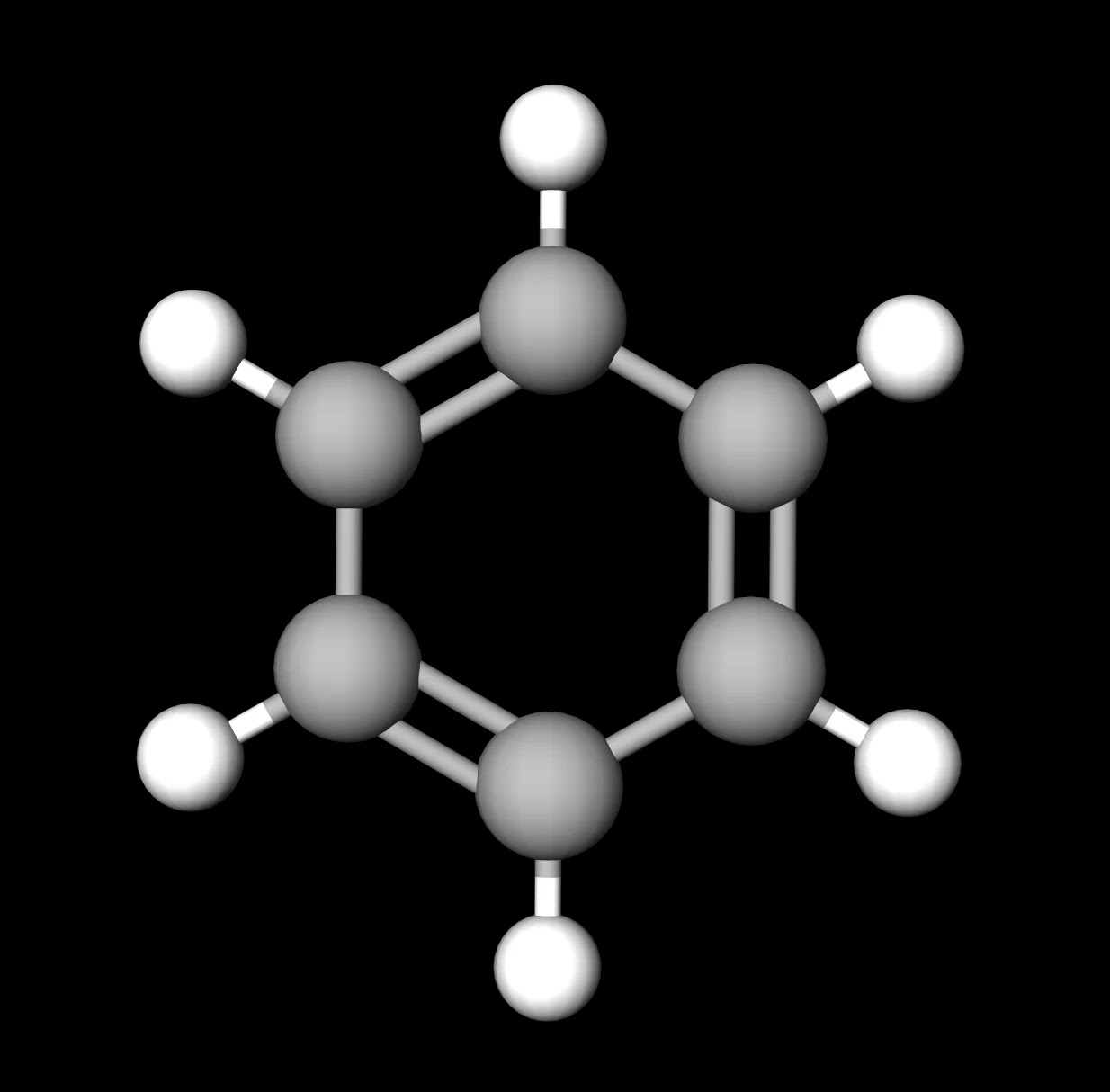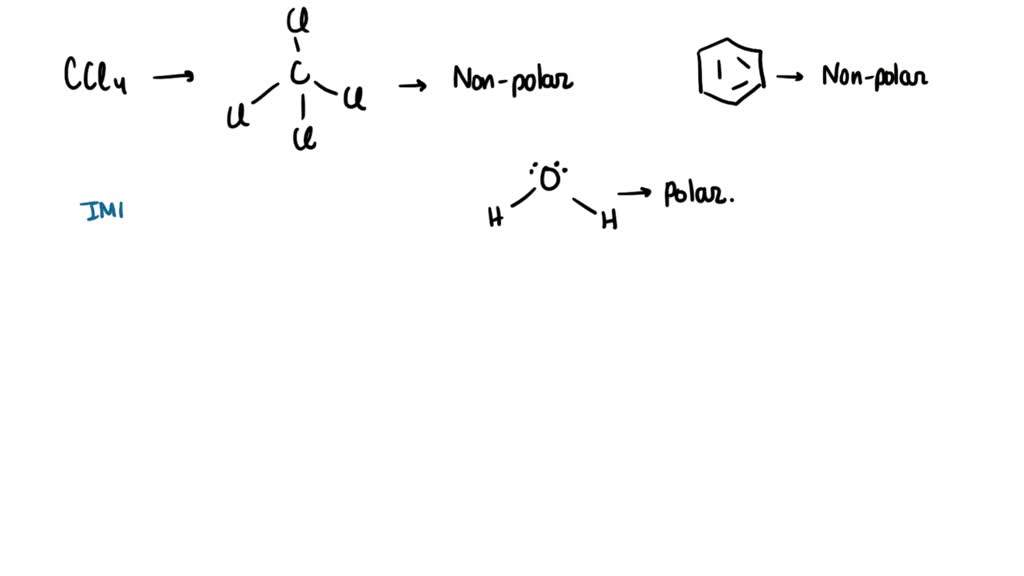
SOLVED: Carbon tetrachloride, CCI4(l), is more mixable with benzene, C6H6(l), than with H2O because (1) the intermolecular forces are similar in both carbon tetrachloride and benzene. (2) both carbon tetrachloride and benzene

text{ }{{\\text{C}}_{\\text{6}}}{{\\text{H}}_{\\text{6}}}\\text{ }$ is a very good industrial solvent for:A) $\\text{ NaCl }$B) $\\text{ MgC}{{\\text{l}}_{\\text{2}}}\\text{ }$C) $\\text{ CaC}{{\\text{O}}_{\\text{3}}}\\text{ }$D) Fats