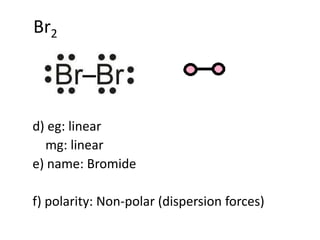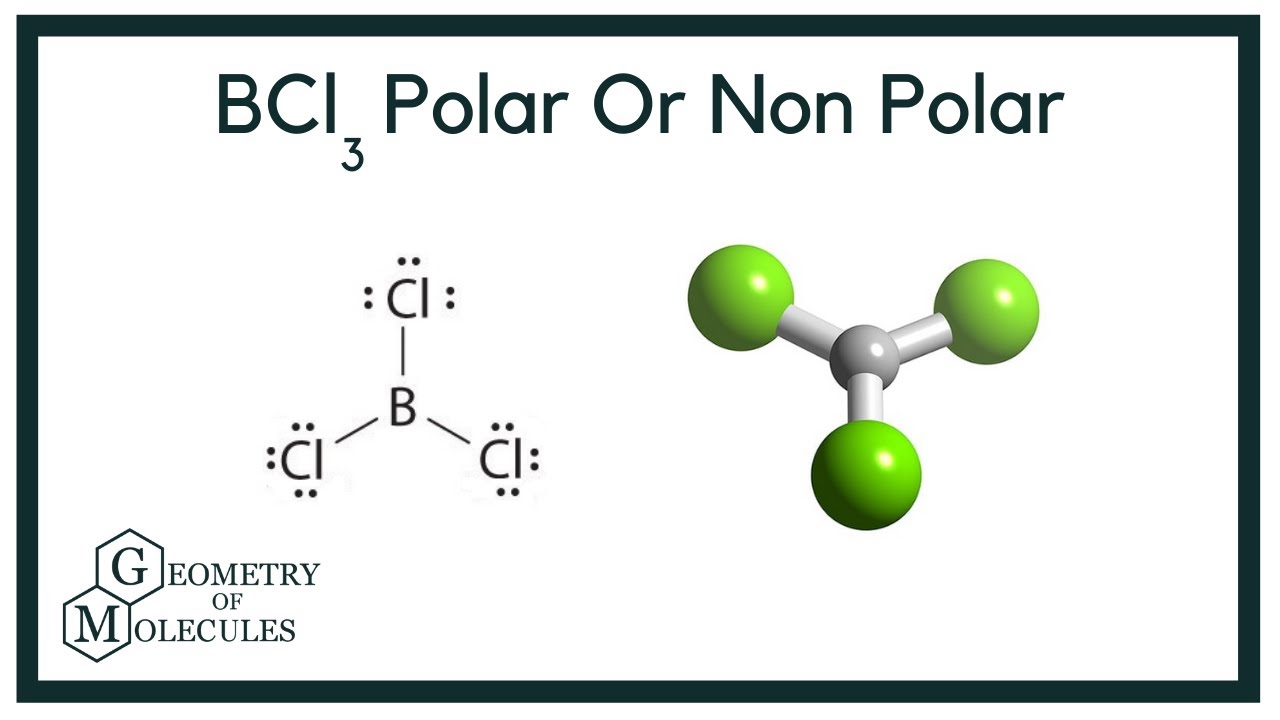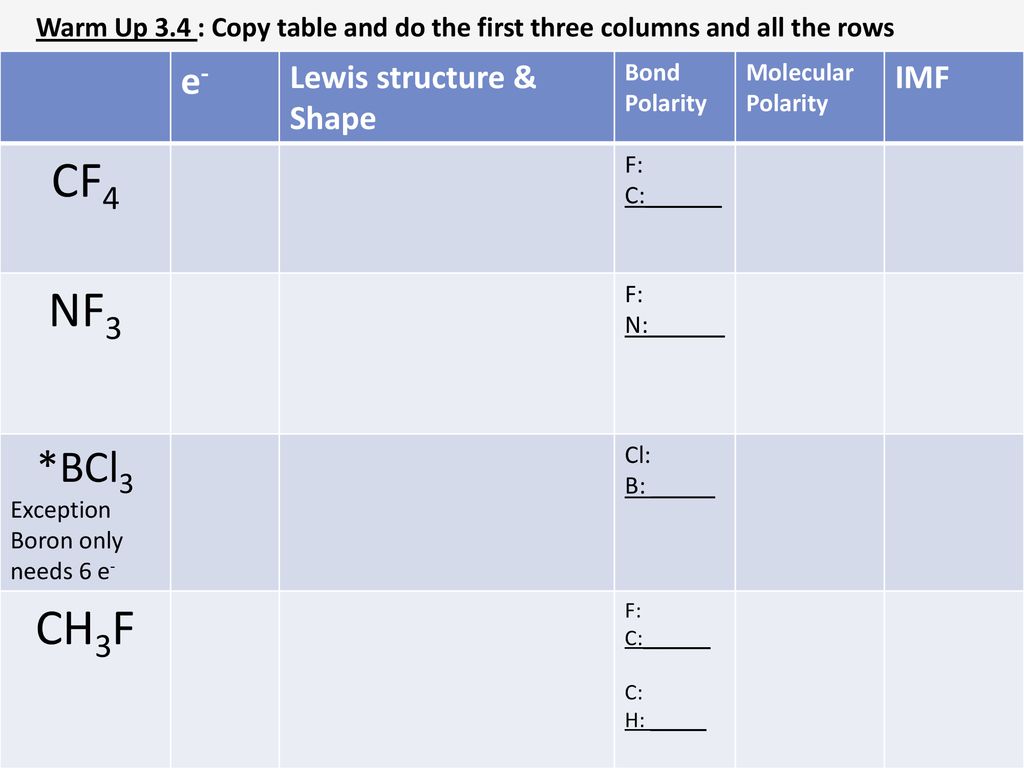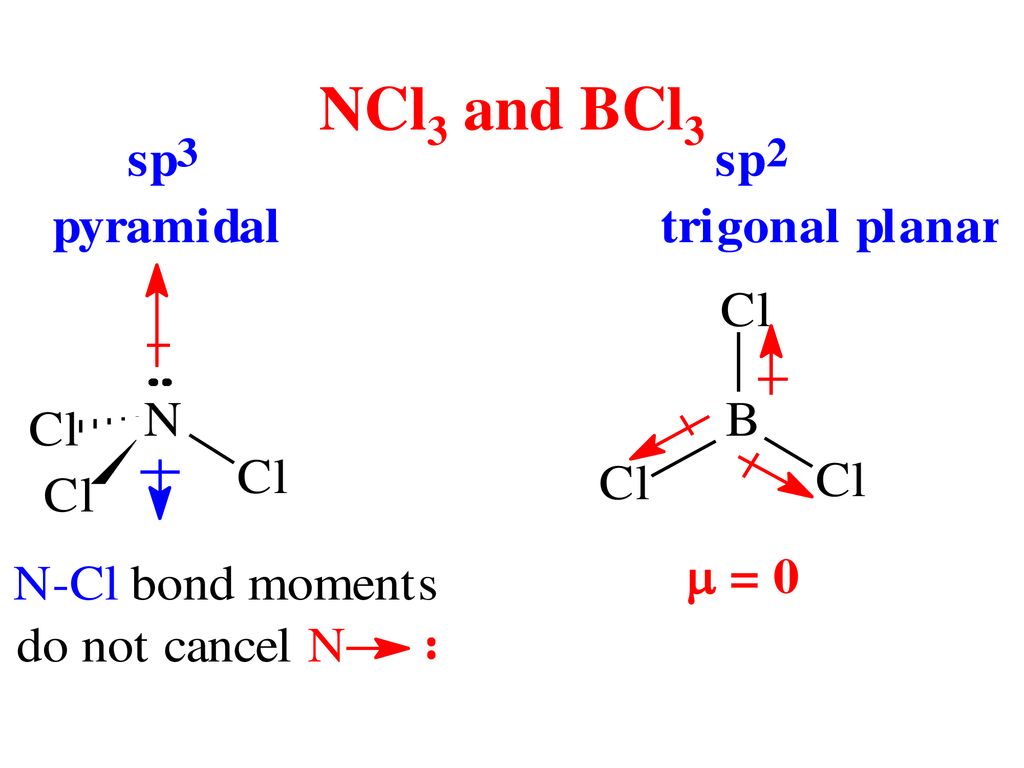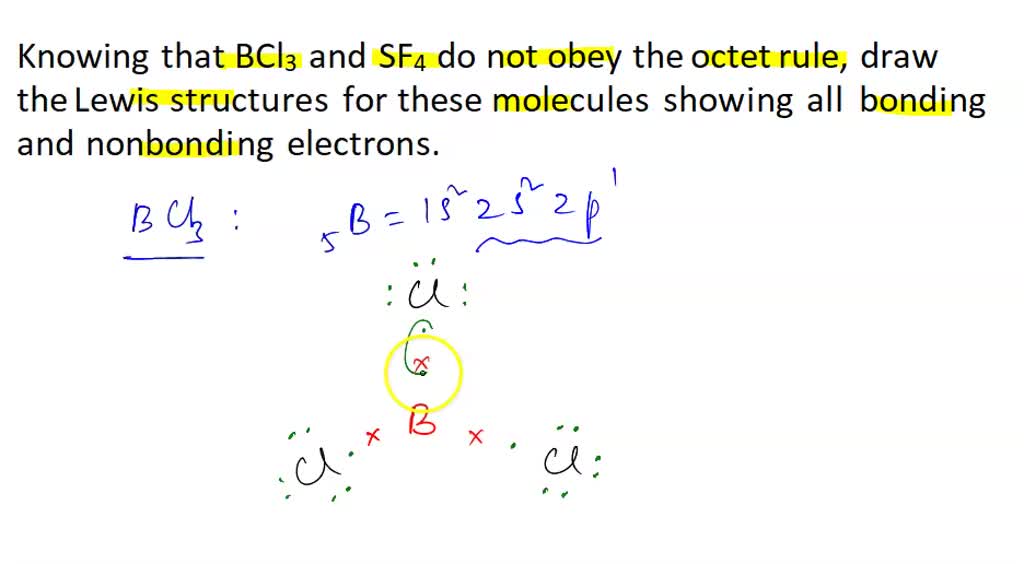
SOLVED: 1)Knowing that BCl3 and SF4 do not obey the octet rule, draw the Lewis structures for these molecules showing all bonding and nonbonding electrons.2)Draw Lewis structures for NH3, CCl4, and N2.

BCl3 lewis structure, molecular geometry, polar or nonpolar, hybridization, Bond angle | Molecular geometry, Molecular, Vsepr theory